First and only oral therapy approved for mild, moderate, and severe plaque psoriasis, and active PsA SEE THE DATA

Plaque Psoriasis
OTEZLA:
4 INDICATIONSOtezla® (apremilast)/Otezla XR™ (apremilast) is indicated for the treatment of adult patients with plaque psoriasis who are candidates for phototherapy or systemic therapy.Read more
*Estimates of patients treated reflect global data since launch (Apr 2014-Mar 2023; US=59% of data). Calculations based on observed drug utilization parameters and number of units distributed. Utilization patterns change over time to best represent current markets.
FDA, U.S. Food and Drug Administration; PsA, psoriatic arthritis; TB, tuberculosis.
References: 1. Otezla [package insert]. Thousand Oaks, CA: Amgen Inc. 2. Data on file, Amgen Inc. 3. Otezla® (apremilast) FDA approval letter. March 21, 2014.
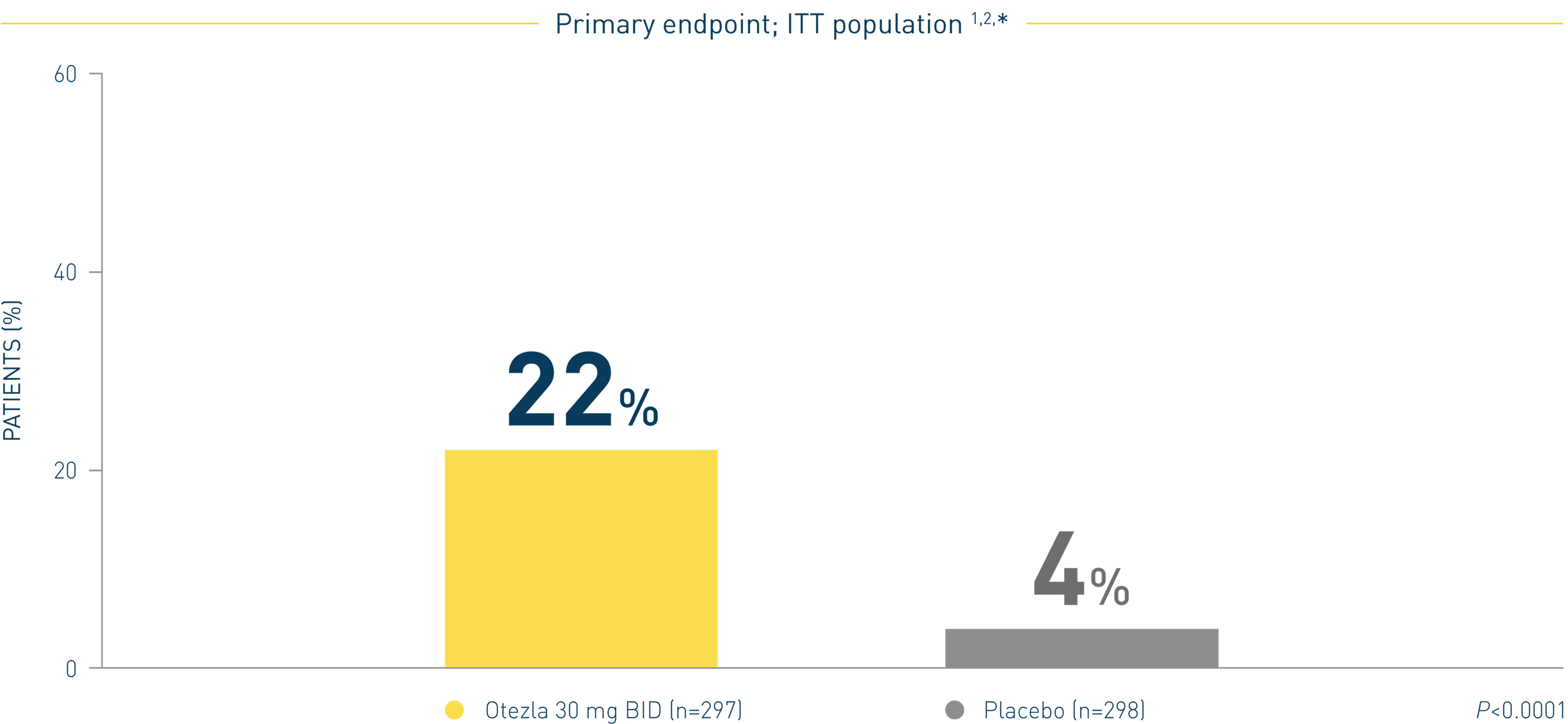
*sPGA response was defined as the percentage of patients who achieved an sPGA score of 0 (clear) or 1 (almost clear) and a ≥2-point reduction from baseline. 2


†In patients with ScPGA ≥2 at baseline. ScPGA score of clear [0] or almost clear [1] with at least a 2-point reduction from baseline.

Analyses at week 2 are exploratory and have not been adjusted for multiple comparisons. No statistical or clinical conclusions can be drawn.
‡In patients with WBI-NRS ≥4 at baseline. §P<0.0001.
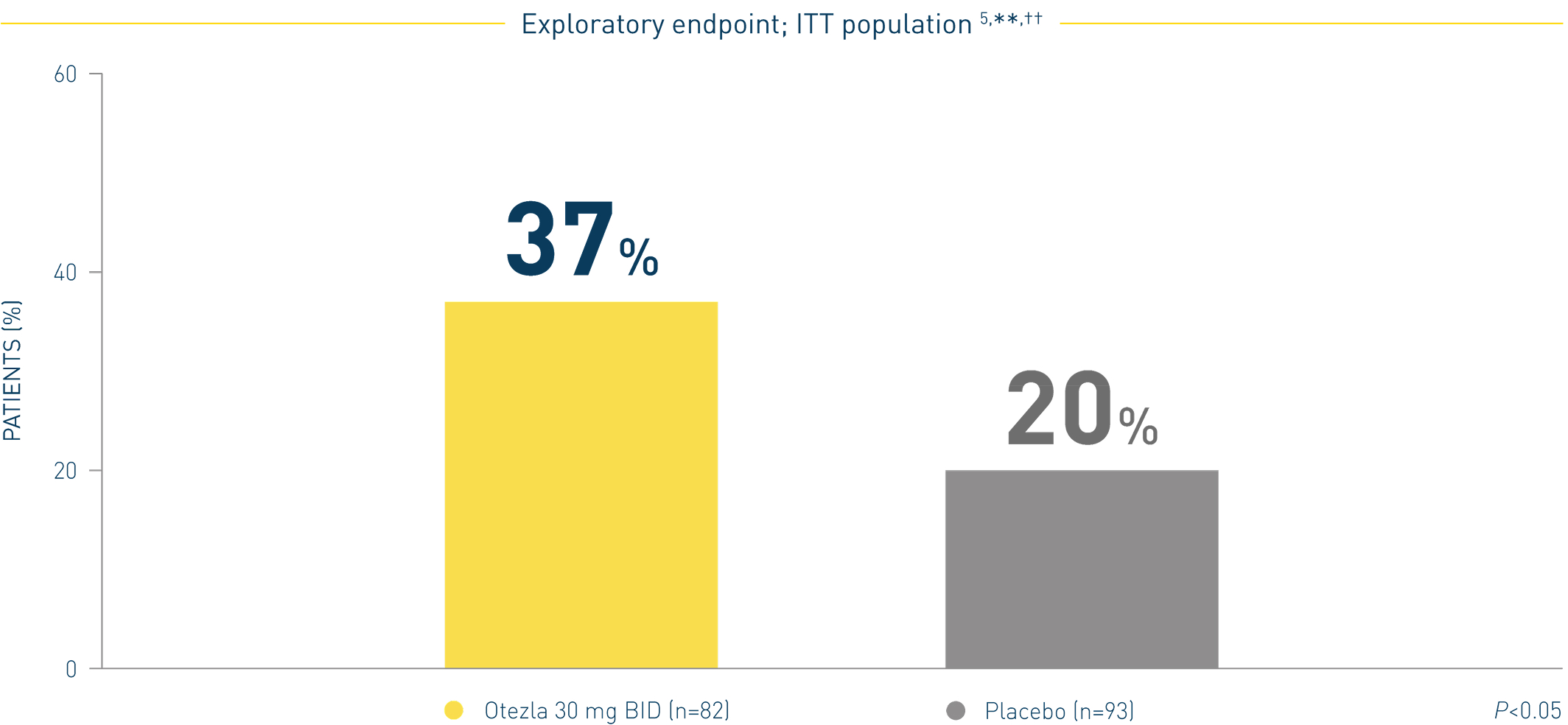
**In patients with NAPSI score >0 at baseline. ††Analysis is exploratory and has not been adjusted for multiple comparisons. No conclusions of statistical significance can be drawn.
BID, twice daily; BSA, body surface area; ITT, intent to treat; MI, multiple imputation; NAPSI, Nail Psoriasis Severity Index; PASI, Psoriasis Area and Severity Index; ScPGA, Scalp Physician Global Assessment; sPGA, static Physician Global Assessment; WBI, whole body itch; WBI-NRS, whole body itch numeric rating scale.
Contraindications
Otezla/OTEZLA XR is contraindicated in patients with a known hypersensitivity to apremilast or to any of the excipients in the formulationWarnings and Precautions
Hypersensitivity: Hypersensitivity reactions, including angioedema and anaphylaxis, have been reported during postmarketing surveillance. If signs or symptoms of serious hypersensitivity reactions occur, discontinue Otezla/OTEZLA XR and institute appropriate therapyContraindications
Warnings and Precautions
Adverse Reactions
Use in Specific Populations
Please click here for the full Prescribing Information.
References: 1. Otezla [package insert]. Thousand Oaks, CA: Amgen, Inc. 2. Stein Gold L, Papp K, Pariser D, et al. J Am Acad Dermatol. 2022;86(1):77-85. 3. Van Voorhees AS, Feldman SR, Lebwohl MG, et al. The Psoriasis and Psoriatic Arthritis Pocket Guide. psoriasis.org/the-pocket-guide/ 4. Schafer P. Biochem Pharmacol. 2012;83(12):1583-1590. 5. Data on file, Amgen Inc.