First and only oral therapy approved for mild, moderate, and severe plaque psoriasis, and active PsA SEE THE DATA

Plaque Psoriasis
OTEZLA:
4 INDICATIONSOtezla® (apremilast)/Otezla XR™ (apremilast) is indicated for the treatment of adult patients with plaque psoriasis who are candidates for phototherapy or systemic therapy.Read more
*Estimates of patients treated reflect global data since launch (Apr 2014-Mar 2023; US=59% of data). Calculations based on observed drug utilization parameters and number of units distributed. Utilization patterns change over time to best represent current markets.
FDA, U.S. Food and Drug Administration; PsA, psoriatic arthritis; TB, tuberculosis.
References: 1. Otezla [package insert]. Thousand Oaks, CA: Amgen Inc. 2. Data on file, Amgen Inc. 3. Otezla® (apremilast) FDA approval letter. March 21, 2014.
Please read the Prescribing Information for instructions on how to administer Otezla/Otezla XR.

*Once-daily dosing starts after titration.
†Otezla tablets should be used for the initial titration regardless of whether Otezla or Otezla XR will be used for the maintenance dosage.
‡Creatinine clearance (CLcr) <30 mL/min estimated by the Cockcroft-Gault equation.
§Otezla XR is not recommended for adult patients with severe renal impairment; the appropriate dosage for these patients has not been determined.

**Pediatric patients, 6 years of age or older, with moderate to severe plaque psoriasis, and weighing at least 20 kg.
††Otezla tablets should be used for the initial titration regardless of whether Otezla or Otezla XR will be used for the maintenance dosage.
Titration of Otezla/Otezla XR is intended to reduce the gastrointestinal symptoms associated with initiation of therapy 1
For pediatric patients with severe renal impairment: 1,‡‡,§§
For initial dosage titration, it is recommended that Otezla be titrated using only the AM schedule shown above for appropriate body weight category and that the PM doses be skipped. 1Recommended maintenance dosage is also based on body weight:
‡‡Creatinine clearance (CLcr) <30 mL/min estimated by the Cockcroft-Gault equation.
§§Otezla XR is not recommended for pediatric patients with severe renal impairment; the appropriate dosage for these patients has not been
determined.
BID, twice daily; QD, once daily.
Provide the 14-day treatment initiation pack sample to patients before they leave the office or prescribe the 28-day treatment initiation pack through your specialty pharmacy of choice.
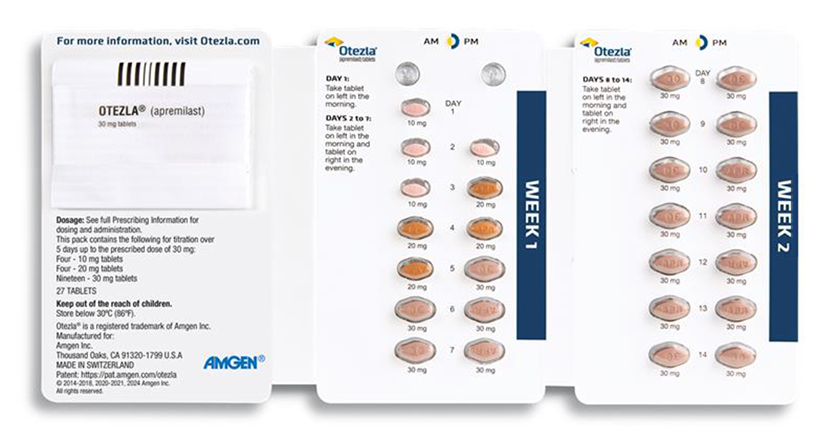
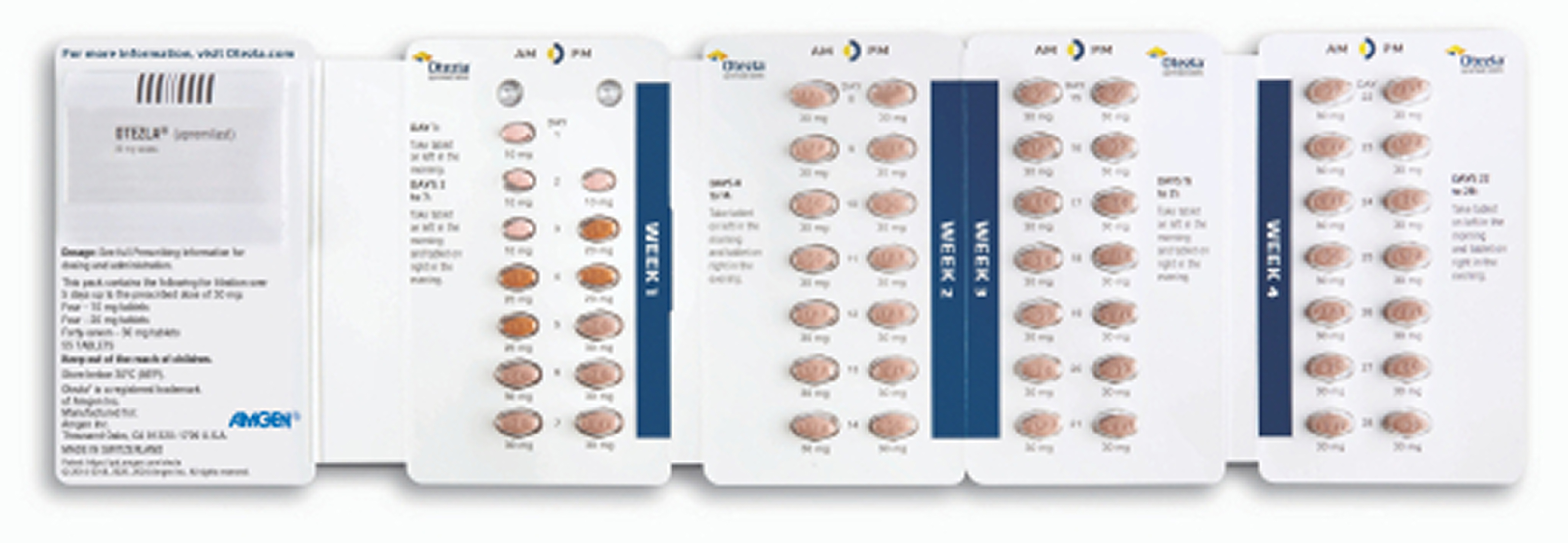

***Otezla was studied over 5 years in adults with psoriatic arthritis and moderate to severe plaque psoriasis.
Contraindications
Otezla/OTEZLA XR is contraindicated in patients with a known hypersensitivity to apremilast or to any of the excipients in the formulationWarnings and Precautions
Hypersensitivity: Hypersensitivity reactions, including angioedema and anaphylaxis, have been reported during postmarketing surveillance. If signs or symptoms of serious hypersensitivity reactions occur, discontinue Otezla/OTEZLA XR and institute appropriate therapyContraindications
Warnings and Precautions
Adverse Reactions
Use in Specific Populations
Please click here for the full Prescribing Information.
References: 1. Otezla [package insert]. Thousand Oaks, CA: Amgen Inc. 2. Data on file, Amgen; 2023. 3. Yélamos O, Ros S, Puig L. Psoriasis (Auckl). 2015;5:109-115. 4. Hugtenburg JG, Timmers L, Elders PJ, et al. Patient Prefer Adherence. 2013;7:675-682. 5. Data on file, Amgen; 2021. 6. Paul C, Cather J, Gooderham M, et al. Br J Dermatol. 2015;173(6):1387-1399. 7. Van Voorhees AS, Feldman SR, Lebwohl MG, et al. The Psoriasis and Psoriatic Arthritis Pocket Guide. psoriasis.org/the-pocket-guide/